New Way to Help Aging Cells Produce Collagen
- This treatment significantly reduced wrinkles in mice.

Scientists have demonstrated that extracellular vesicles loaded with mRNA coding for collagen production can be easily produced and delivered into aging fibroblasts in vitro and in vivo, boosting collagen levels and eliminating signs of skin aging in mice [1].
Superior delivery method
Finding a good way to deliver molecular cargo into cells is important. Some novel therapies, such as the mRNA-based SARS-CoV2 vaccines, utilize lipid nanoparticles (LNPs) to solve the problem of mRNA’s immunogenicity and instability. However, LNPs are not without their own issues: they too can be immunogenic as well as cytotoxic, and they are often not specific enough with regard to target cell types.
Extracellular vesicles (EVs) can be a good alternative to LNPs. EVs are tiny droplets that, in nature, “bud off” the cellular membrane to facilitate cell-to-cell communication, shuttling various molecules between cells without causing an immune reaction. However, creating customized vesicles loaded with specific molecular cargo is not easy.
Packed with collagen
In this new study published in Nature, the researchers were able to generate vesicles loaded with mRNA that encodes for collagen I alpha I (COL1A1). Age-related collagen depletion leads to degradation of the extracellular matrix and skin aging [2]. Many products and procedures claim to help with collagen deficiency, but as the authors note, none of the currently available technologies has been effective in achieving long-term collagen replacement.
The same group of researchers previously developed a method of EV production called cellular nanoporation (CNP) [3]. Using it, they created similar EVs that were then introduced into aged fibroblasts, cells that produce collagen proteins. EVs generated by the new method contained more than 200 times the amount of COL1A1 mRNA than EVs produced by the more conventional bulk electroporation (BEP) and 3,000 times more than EVs naturally secreted by control cells.
The researchers then treated cultured fibroblasts with COL1A1-EVs for 48 hours. Interestingly, this increased not just collagen content but also proliferation of fibroblasts. Levels of pro-collagen I, a precursor of COL1 protein, were significantly increased following COL1A1-EV treatment, showing a spike in endogenous collagen production.
Wrinkles be gone
For their in vivo experiments, the researchers used a mouse model of UV-induced acute photoaging that closely mimics skin aging in humans, complete with lots of wrinkles. They pitted their EVs against healthy controls that had not been irradiated and a group that had been treated with collagen-containing LNPs.
COL1A1 mRNA became significantly elevated in local skin tissue, peaking at 12 hours after the EV delivery and returning to baseline levels after four days. Both LNP and EV treatments led to decreases in wrinkle number and area, but in the EV group, the effect was much more pronounced. By day 28, the EV group had as few wrinkles as the healthy controls. Skin treated with COL1A1-EVs and COL1A1-LNPs also showed higher elasticity and firmness.
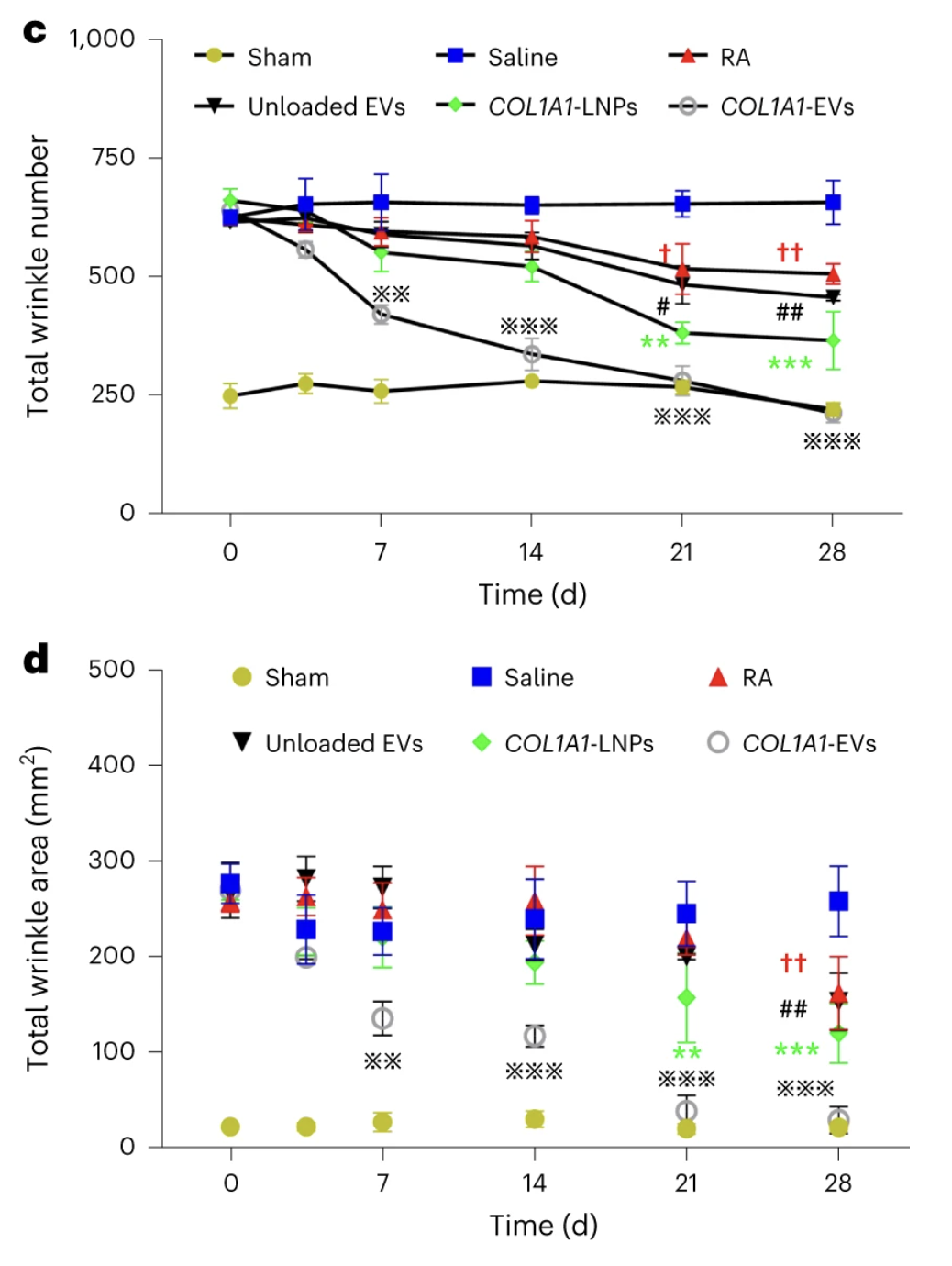
A subset of mice was monitored for another month. Unfortunately, wrinkles reappeared about one week later, and at the end of the follow-up period (almost two months after the treatment), the wrinkle number and area became indistinguishable from pre-treatment levels.
The researchers also compared the immunogenic side effects of LNPs and EVs. 24 hours after injection, skin treated with COL1A1-LNPs showed redness, swelling, recruitment of immune cells, and high levels of the inflammatory cytokines TNF-α, IL-6, IL-1β, and IFN-γ. Conversely, tissue treated with COL1A1-EVs did not exhibit a strong inflammatory reaction, showing that EVs are much less immunogenic than LNPs.
Microneedles and repeated treatments
The researchers then repeated their experiments with a different method of applying EVs. Instead of injection, they used microneedle patches treated with EVs. Such patches consist of a base and a layer of microneedles. After application, the base is removed, and the microneedles dissolve in the skin along with their cargo.
Analysis showed that while subcutaneous needle injection resulted in uneven delivery of EVs and clumping, EVs delivered by microneedle were better dispersed. The microneedle method was also more effective, causing the reduction in wrinkles to linger twice as long compared to injection.
Finally, to see whether collagen replacement and wrinkle reduction could be maintained for longer, the researchers subjected mice to monthly treatments, which continued to be effective without causing any side effects. The microneedle treatment also proved superior here.
Conclusion
These results show that boosting endogenous collagen production through mRNA might be able to, at least temporarily, ameliorate skin wrinkling with age. This proof-of-concept study delivers encouraging results and showcases two promising technologies with high translational potential: mRNA delivery through CNP-created EVs and microneedles for EV application.
Literature
[1] You, Y., Tian, Y., Yang, Z. et al. Intradermally delivered mRNA-encapsulating extracellular vesicles for collagen-replacement therapy. Nat. Biomed. Eng (2023).
[2] Sparavigna, A. (2020). Role of the extracellular matrix in skin aging and dedicated treatment-state of the art. Plastic and Aesthetic Research, 7, 14.
[3] Yang, Z., Shi, J., Xie, J., Wang, Y., Sun, J., Liu, T., … & Lee, L. J. (2020). Large-scale generation of functional mRNA-encapsulating exosomes via cellular nanoporation. Nature biomedical engineering, 4(1), 69-83.










