Metformin: Benefits, Side Effects, and Research
There is a significant amount of enthusiasm for the off-label use of metformin in the longevity community. We take a look at this drug in the context of aging, the gut microbiome, and the other ways in which it influences our biology.
What is metformin?
Metformin is a drug in the biguanide class used for the treatment of type 2 diabetes. It was discovered in 1922 and Metformin was originally isolated from naturally occurring molecules found in the plant Galega officinalis, known as French lilac. French physician Jean Sterne began the first scientific study in humans in the 1950s and it was approved as a medication in France in 1957. Perhaps somewhat surprisingly it was not approved in the United States until 1995!
Metformin is widely prescribed for type 2 diabetes; it improves glucose metabolism and thereby reduces diabetic complications. How it works is not well-understood, but we know that metformin mostly interacts with signaling molecules in the liver, gut, and kidney. Results vary according to the dosage and duration of metformin treatment. Metformin has multiple mechanisms of action, which likely account for its anticystic, antitumor, cardioprotective, neuroprotective, and longevity effects.
Metformin and glucose metabolism
Diabetes is an age accelerator. Compared to non-diabetic individuals, the phenotypic age (PhAge) of individuals with type 1 and type 2 diabetes is 12.02 and 16.32 years higher, respectively [1]. Metformin helps normalize glucose levels and therefore may slow changes in PhAge.
Metformin’s journey in the body begins with gut cells called enterocytes. Metformin carries a positive charge, which makes it relatively easy to cross the negatively charged cell membrane. Further, metformin is aided by the OCT1 receptor in the cell membrane.
Once inside the cell, metformin makes it way to mitochondria. The matrix side of the inner mitochondrial membrane is also negatively charged, and this allows metformin to accumulate inside mitochondria in quantities 1000 times greater than outside the cell [2].
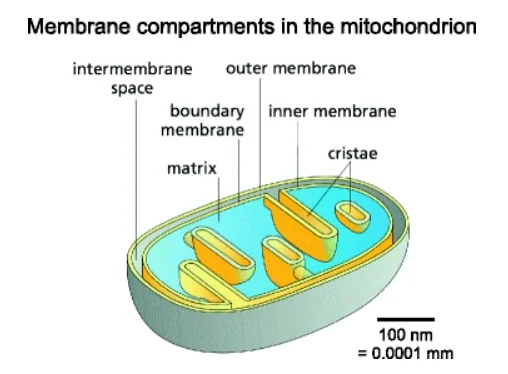
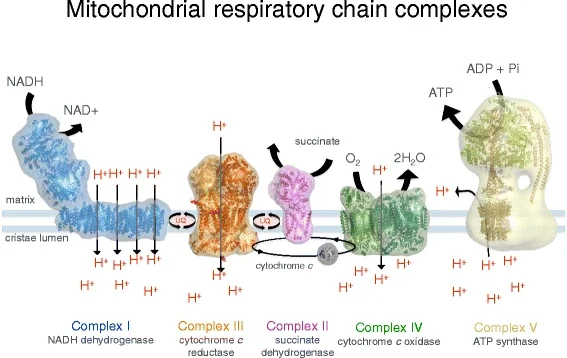
Within mitochondria, metformin directly inhibits the function of complex 1, a cluster of proteins vital to the function of the electron transport chain. This results in a dampening of the production of ATP using oxygen (aerobic respiration) in the cell.
The cell, in this instance, must depend on anaerobic glycolysis (making energy from glucose without oxygen). Not only does this use up a lot of glucose inefficiently, it also produces a lot of lactic acid as a byproduct. Excessive lactic acid can lead to metabolic acidosis, which can be a serious side effect of metformin use in certain contexts.
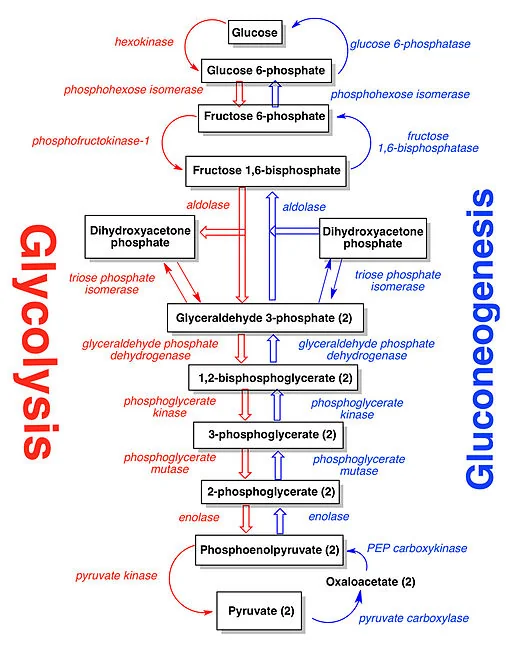
The lactate is transported to the liver, where it is subjected to the Cori cycle. The Cori cycle normally converts lactic acid to pyruvate. Pyruvate, in turn, can be made into glucose by entering the Krebs cycle [3]. The energy required to operate the Cori cycle further reduces available glucose. However, this is not where the story ends.
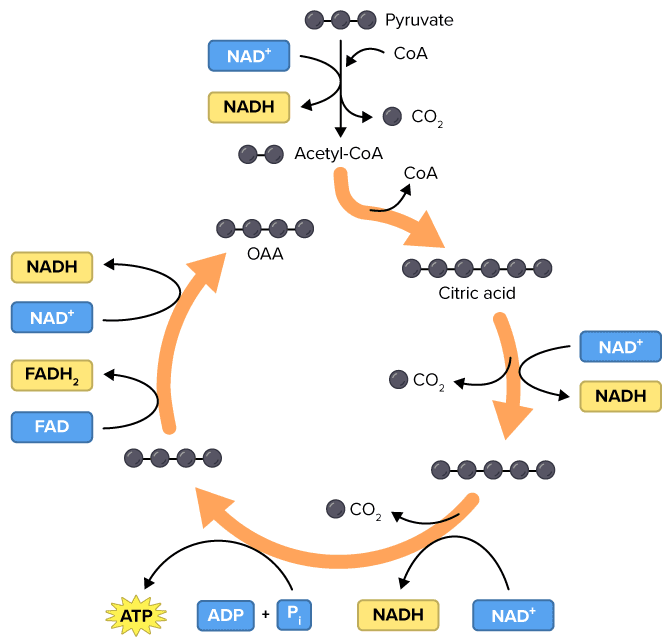
Metformin also blocks the action of pyruvate carboxylase [3]. Pyruvate carboxylase catalyzes the conversion of pyruvate to oxaloacetate (OAA) in the Krebs cycle. This puts the kibosh on ATP production and causes a buildup of pyruvate that gets converted back to lactate with the aid of the catalyst lactate dehydrogenase. This can result in metabolic acidosis.
As an interesting aside, NADH is used up in this process, reducing the ratio of NAD+ to NADH, which could, in theory, trigger the expression of sirtuins [4].
As ATP production plummets in the cell, the ratio of ATP to AMP is reduced, which triggers the nutrient sensing molecule AMPK to actively downregulate the synthesis of fat, increase the use of fat for energy, and increase insulin sensitivity. This may seem trivial as it primarily involves the lining of the small intestine, but the area of the small intestine is about the size of a tennis court.
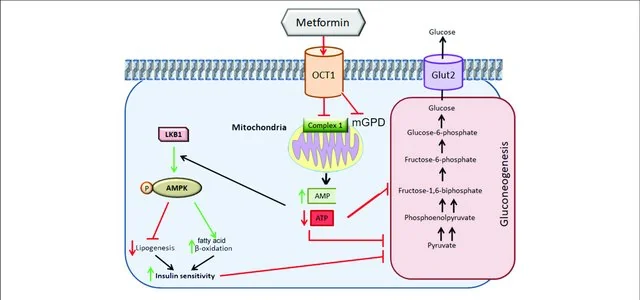
Furthermore, enterocytes have a voracious appetite. The intestine and its accessory organs account for about 6% of our total bodyweight but 20-25% of our total energy expenditure [6]. Finally, the concentration of metformin is 100-300 times higher in the intestine than it is in serum [7]. This suggests that mitochondria in enterocytes are the primary targets of metformin and that the effect of metformin on fat burning and glucose availability is very significant.
In sum, metformin’s entry into intestinal cells causes the intestine and liver to use glucose inefficiently, burn more fat, and increase insulin sensitivity in these areas. Metformin’s action in the intestine is not limited to its interaction with enterocytes, as it also interacts with the gut microbiome.
Metformin and the gut microbiome
Evolutionary theory tells us that at some point in the distant past, primitive aerobic bacteria were engulfed by primordial anaerobic cells. Over time, this symbiotic relationship evolved, and these aerobic bacteria became what we refer to today as mitochondria. With this in mind, we might expect gut bacteria, especially aerobic gut bacteria, to be affected by metformin in similar ways as enterocytes.
A significant number of recent studies have focused on how metformin affects gut microbiota, which affect glucose metabolism. Irrespective of metformin, gut microbiota do affect glucose metabolism in several ways. In the presence of metformin, gut bacteria appear to better maintain the integrity of the intestinal barrier, promote the production of short-chain fatty acids, regulate bile acid metabolism, and improve glucose homeostasis, independent of other factors [7].
Metformin inhibits the normal increase in lipopolysaccharide (LPS) seen in serum following the ingestion of a high-fat meal. LPS is a toxin derived from the cell walls of bacteria. A decrease in LPS suggests that either the composition of gut microbiota has changed or that the intestinal barrier has been modified in a way that blocks normal access by LPS into the bloodstream [7].
Short-chain fatty acids (SCFAs) are produced in the gut because of the bacterial fermentation of fiber [8]. 80% of these SCFAs are either acetate, butyrate, or propionate [9]. Metformin increases the number of SCFA-producing bacteria in animal studies. Similar findings were reported in people with type 2 diabetes who take metformin [7]. SCFAs improve insulin sensitivity in fat, muscle, and liver tissue by controlling fat, glucose, and amino acid metabolism and function.
SCFAs also stimulate the secretion of (GLP-1) in intestinal epithelial cells [10]. GLP-1 slows down glucose spikes after eating, and GLP-1 agonists improve the ability of insulin-producing cells in the pancreas to maintain blood sugar levels right after a meal [11]. The blockbuster diabetes drug Ozempic is a GLP-1 agonist, which is now under review by the FDA as an anti-obesity drug for non-diabetic individuals [12].
In addition, SCFAs produce wide-ranging effects on the function of the human body that go well beyond glucose metabolism. For example, SCFAs are an integral part of the gut-brain axis, a system that enables “crosstalk” between the microbiome and the brain [13,14].
Metformin has significant effects on the regulation of bile acid metabolism. Studies suggest that metformin may improve glucose metabolism by regulating total bile acid in the serum. In a study of diabetic rats, bile salt hydrolase produced by gut microbiota increased in the second month of metformin administration.
Metformin also increased the level of GUDCA (a bioactive acid) by regulating gut microbiota. Specifically, GUDCA was increased by inhibiting the growth of certain bacterial species, which inhibited the FXR signaling pathway to reduce blood glucose and maintain blood glucose homeostasis. Metformin also reduced the reabsorption of bile in the distal ilium, leading to increased bile salt concentrations within the colon. This may have accounted for the effects of metformin on the colonic microbiota. This same mechanism could, in theory, reduce both serum cholesterol and LPS levels [15,16].
The nuclear receptor FXR functions as the chief sensor of bile acid levels and the main executor of bile acid-induced gene expression [17]. Bile acids have been shown to activate the FXR receptor and subsequently alter the expression of genes involved in the making of new glucose in the body (gluconeogenesis). These genes include PEPCK, G-6-P, and fructose-1-6-biphosphate [18].
Metformin increases the abundance of specific members of the microbiome, which improve barrier function in the intestine. Metformin increases the abundance of Akkermansia muciniphila. Low concentrations of this microbe may indicate glucose intolerance [19]. Akkermansia muciniphila consumes a great deal of mucins, which are very heavy molecules made of protein and sugar.
In combination with antimicrobial peptides, mucins in epithelial tissues make mucus. This keeps cells hydrated and provides an immunoprotective barrier to would-be pathogens. Akkermansia muciniphila’s presence is negatively correlated with obesity, diabetes, and cardiovascular disease, and it helps to reduce the migration of LPS into the bloodstream and improves blood glucose levels [20].
Once metformin is absorbed in the gut, it makes its way to the liver. In the liver, metformin lowers hepatic glucose production (gluconeogenesis) and insulin sensitivity [2,21].
As with enterocytes’ mitochondria, metformin also accumulates in the mitochondria of liver cells (hepatocytes). This occurs over a period of hours and inhibits the operation of the electron transport chain, which reduces ATP production. This would logically result in a change in the ratio of ATP to AMP; however, this finding has failed to consistently materialize in studies [22,23].
Evidence suggests another way that metformin could inhibit gluconeogenesis is by the inhibition of mitochondrial glycerophosphate dehydrogenase (mGPD) in the liver. mGPD is a component in one of two systems that allow NADH to be made in the cytosol and able to contribute to the Krebs cycle to generate ATP [24].
It is not clear which of these inhibitory mechanisms that stem from metformin administration has the greater impact on gluconeogenesis in the liver.
When the ratio of ATP to ADP or AMP goes down sufficiently, AMPK is activated, which triggers catabolic activity to generate more ATP and shuts down other metabolic activity that unnecessarily consumes ATP [25]. Metformin does not rely exclusively on the inhibition of complex 1 in mitochondria to activate AMPK. To some extent, metformin can also activate AMPK via a lysosomal pathway [26].
Metformin’s effects on glucose production in the liver can be dependent on or independent of AMPK. Evidence suggests that metformin can downregulate gluconeogenesis by altering the gene expression of enzymes such as PEPCK and G6Pase [27]. Evidence also indicates that AMPK modulates fructose-1-6 biphosphatase, a key enzyme of gluconeogenesis.
Changes in the ratio of ATP to AMP trigger AMPK. AMP also builds up because of the consumption of ATP. When this occurs, AMP directly inhibits adenylate cyclase, which inhibits the expression of enzymes associated with gluconeogenesis [28,29].
A major long-term effect of metformin is the enhancement of hepatic insulin sensitivity. Mouse studies suggest that this is also mediated by AMPK. AMPK inhibits fat synthesis and activates fat oxidation in the liver by direct phosphorylation of the two isoforms of acetyl CoA carboxylase (ACC1/ACC2).
If this phosphorylation does not occur for some reason, fat synthesis is increased, fat oxidation is decreased, and hyperglycemia, hyperinsulinemia, glucose intolerance, and insulin resistance follow, even on a regular diet [30]. What this boils down to is that metformin enhances insulin sensitivity, at least in mice, by phosphorylation of ACC1 and ACC2. The long-term insulin-sensitizing effects of metformin appear to be mediated entirely by AMPK.
Polycystic ovary syndrome (PCOS) and metformin
Insulin resistance drives PCOS, which causes female infertility in many instances. Metformin reduces insulin resistance and there may prevent or mitigate the effects of PCOS.
A review of metanalysis, randomized controlled studies, and long-term observational studies indicate that metformin complements traditional treatments for PCOS. This is evidenced by an increase in success rates and a decrease in complication rates during the administration of PCOS-related treatments [34-35].
Diabetes, cancer and metformin
Diabetes increases the risk of colorectal, pancreatic, liver [36], endometrial [37], breast [38], and bladder [39] cancers. Evidence suggests that common factors, such as obesity, systemic inflammation, and smoking, lead to both diabetes and cancer [40]. Additionally, diabetes drives cancer by increasing levels of reactive oxygen species (ROS) and lowering antioxidant capacity. This leads to increased DNA damage.
Metformin can check cancer development in two interrelated ways. First, metformin improves glucose metabolism. This leads to decreased ROS production, improved antioxidant capacity, reductions in serum insulin, and insulin growth factor I (IGF-I), which are potent mitogens for cancer cells [41].
Second, metformin affects signaling pathways that improve DNA repair and slow cell growth, proliferation, and metastasis. When metformin is introduced into the body, ATM and its target Chk2 are immediately activated. ATM is a crucial part of the DNA damage response system. This system not only initiates DNA repair but can also shut down cell division [42]. Therefore, metformin’s activation of ATM helps repair DNA damage, which keeps cells from becoming cancerous and slows down cell division when they do.
Metformin inhibits mTOR activity in a variety of ways. The inhibition of mitochondrial complex I activates AMPK by decreasing the ratio of ATP to AMP. When AMPK becomes active, its effects are not limited to glucose and fat metabolism. Activated AMPK can directly inhibit mTOR [43]. Metformin can also inhibit mTOR through REDD1 [44]. Finally, metformin regulates the PI3K/Akt pathway to dampen mTOR activity as well.
Inhibition of mTOR has complex effects in different tissues. For example, mTOR activity is associated with apoptosis in some cancer cells, which is a good thing, but mTOR inhibition also slows protein synthesis, growth and proliferation of cancer cells [44]. Rapamycin is used in clinical settings to inhibit mTOR and has been shown to have favorable effects on cancer prevention, particularly colon cancer [45-46]. Therefore, metformin may foster many of the same antitumor benefits ascribed to rapamycin.
Metformin, via AMPK and ATM activity, also activates tumor suppressor p53 [42]. Studies show that p53 activation inhibits melanoma invasion, metastasis, and the triggering of cell death in lymphoma cells [47].
Finally, numerous studies suggest that metformin may complement other cancer treatment strategies. If has been used in conjunction with radiotherapy and chemotherapy and to improve the response to treatment in androgen derivatives (ADTs) [36].
Metformin’s cardioprotective effects
Researchers reviewed 40 studies investigating the effects of metformin on cardiac function among 1,066,408 patients with coronary artery disease. Metformin reduced cardiovascular mortality and the number of cardiovascular events (heart attacks, etc.) [48].
Heart attack and CAD patients without T2DM did not appear to benefit from metformin in terms of event reduction. However, metformin has been shown to decrease heart damage and improve survival rates after myocardial infarction. Metformin does this by interfering with the body’s normal response to ischemia (oxygen deprivation) [48].
Metformin prevents heart tissue self-destruction that occurs via the ischemia-reperfusion injury. When oxygen is reintroduced to ischemic heart tissue, a series of events are triggered that results in cell death. Metformin counters these events by increasing AKT, activating the reperfusion injury salvage kinase pathway (RISK) [49], activating AMPK, restoring depleted PGC-1α levels, and increasing nitric oxide levels. AKT and RISK activation prevent mitochondria-triggered cell death, AMPK promotes the breakdown of sugar locally to provide energy, PGC-1α upregulates the production of new mitochondria necessary for cell survival and healing, and nitric oxide reduces vascular inflammation [50,51].
Neuroprotective effect
A 2022 review of population-based studies including 194,792 participants looked at the relationship between metformin use and neurodegenerative disease. Overall, metformin users were 23% less likely than non-metformin users to develop neurodegenerative disease. Participants who took metformin for more than 4 years were 71% less likely to develop neurodegenerative disease. The research group noted, however, that there was significant variation between study outcomes and suggested further studies to confirm these findings [52].
Longevity effects
Metformin’s health and apparent longevity benefits have spurred ongoing interest in its use as a longevity drug. Clinical trials, including MILES (Metformin In Longevity Study) and TAME (Targeting Aging with Metformin), were designed for this purpose.
Early results from MILES suggest that metformin may induce changes in gene expression that likely contribute to increased longevity. However, it is not clear if these benefits extend to non-diabetic individuals.
There is still reason for optimism. The TRIIM study published in 2019 combined metformin, DHEA, and HGH. The aim of this study was to regenerate the thymus gland [53]. In doing so, it was hoped that the age-related depletion of T-cells could be mitigated, which would be expected to reduce immunosenescence-related mortality.
Remarkably, after nine months, the fat in the thymus gland of most participants was significantly reduced and imaging data indicated that where fat had been, new thymus tissue was generated. Further T-cell production and function improved, and an array of epigenetic clocks demonstrated for the first time that an anti-aging intervention reduced biological age by 2.5 years in just nine months.
In this study, metformin in combination with DHEA were co-administered to counter the unwanted HGH effects: decreased insulin sensitivity and increased insulin resistance. Although this study does not suggest that metformin independently increases epigenetic age, it does argue for its efficacy in combination with DHEA and GH [53]. At present, the follow-up to the TRIIM study (TRIIM-X) is currently underway and is slated for completion in November 2022.
Despite reason for optimism, the evidence in support of metformin is far from unequivocal. It is not clear that metformin increases maximum lifespan in human beings, although it does appear to reduce early mortality associated with many of the diseases of middle age.
Available evidence suggests that the beneficial effects of metformin on aging and healthspan result primarily from its effects on glucose metabolism, although this evidence is not yet conclusive. These effects include enhanced insulin sensitivity, oxidative stress reduction, and cardioprotection [54,55].
Safety
Taking metformin can cause mild side effects, including diarrhea, nausea, stomach pain, heartburn, and gas. More serious potential side effects include lactic acidosis and hypoglycemia (low blood sugar). You should call your doctor if you experience any side effects while taking metformin [56].
However, metformin has a number of known interactions with other drugs, including diabetes drugs such as insulin and glyburide [57] along with blood pressure management drugs such as furosemide and hydrochlorothiazide [58].
Cholesterol-modifying drugs, such as the NAD+ precursor niacin, are worth noting, as this is a popular supplement in the life extension community and can make metformin less effective in lowering blood sugar levels [59].
Lactic acidosis is an uncommon but very serious side effect of metformin. This is when lactic acid builds up in the bloodstream and is a medical emergency that requires treatment in the hospital. It is fatal in about half of people who develop it [3].
Using metformin with the glaucoma drugs acetazolamide, brinzolamide, dorzolamide, and methazolamide may increase the risk of lactic acidosis [60]. Metformin has known interactions with topiramate, a drug used to treat nerve pain and seizures, and may also raise the risk of lactic acidosis. Phenytoin, which is used to treat seizures, can make metformin less effective in lowering blood sugar. Cimetidine, which is used to treat heartburn and other stomach problems, can interact with metformin and increase the risk of lactic acidosis as well [61].
Some hormone drugs may also make metformin less effective in lowering blood sugar. Corticosteroids like budesonide, fluticasone, prednisone, and betamethasone have potential interactions. Estrogens such as estradiol, conjugated estrogens (Premarin), and birth control pills or patches can also interact. Isoniazid, an anti-tuberculosis drug, can also make metformin less effective in lowering blood sugar [62].
Thyroid drugs may make metformin less effective in lowering your blood sugar too. These include levothyroxine, liothyronine, and liotrix [63].
At therapeutic levels, lactate production is well-controlled and does not pose a threat to health. Under certain circumstances, such as cardiorespiratory failure, sepsis, dehydration, renal impairment, or liver failure, metabolic acidosis can occur. This can be acute or chronic. Acute metabolic acidosis causes massive derangements in metabolism and death. Chronic metabolic acidosis can produce symptoms of nausea, vomiting, and rapid respiration while slowly degrading muscle tissue, dissolving bone, reducing serum albumin, driving chronic kidney disease, stimulating inflammation, impairing both insulin secretion and responsiveness, and causing an increase in the rate of amyloid accumulation [64].
Literature
[1] N. Bahour, B. Cortez, H. Pan, H. Shah, A. Doria, and C. Aguayo-Mazzucato, “Diabetes mellitus correlates with increased biological age as indicated by clinical biomarkers,” GeroScience, vol. 44, no. 1, pp. 415–427, Feb. 2022
[2] G. Rena, D. G. Hardie, and E. R. Pearson, “The mechanisms of action of metformin,” Diabetologia, vol. 60, no. 9, pp. 1577–1585, 2017
[3] V. Rajasurya, H. Anjum, and S. Surani, “Metformin Use and Metformin-associated Lactic Acidosis in Intensive Care Unit Patients with Diabetes,” Cureus, vol. 11, no. 5, pp. e4739–e4739, May 2019
[4] K. A. Anderson, A. S. Madsen, C. A. Olsen, and M. D. Hirschey, “Metabolic control by sirtuins and other enzymes that sense NAD(+), NADH, or their ratio,” Biochim. Biophys. acta. Bioenerg., vol. 1858, no. 12, pp. 991–998, Dec. 2017
[5] M. Faure et al., “Metformin in Reproductive Biology ,” Frontiers in Endocrinology , vol. 9. 2018
[6] H. Yang, X. Wang, X. Xiong, and Y. Yin, “Energy metabolism in intestinal epithelial cells during maturation along the crypt-villus axis,” Sci. Rep., vol. 6, p. 31917, Aug. 2016
[7] Q. Zhang and N. Hu, “Effects of Metformin on the Gut Microbiota in Obesity and Type 2 Diabetes Mellitus,” Diabetes. Metab. Syndr. Obes., vol. 13, pp. 5003–5014, Dec. 2020
[8] D. Parada Venegas et al., “Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases ,” Frontiers in Immunology , vol. 10. 2019
[9] A. Nogal, A. M. Valdes, and C. Menni, “The role of short-chain fatty acids in the interplay between gut microbiota and diet in cardio-metabolic health,” Gut Microbes, vol. 13, no. 1, pp. 1–24, 2021
[10] A. Koh, F. De Vadder, P. Kovatcheva-Datchary, and F. Bäckhed, “From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites,” Cell, vol. 165, no. 6, pp. 1332–1345, Jun. 2016
[11] J. J. Holst, “The Physiology of Glucagon-like Peptide 1,” Physiol. Rev., vol. 87, no. 4, pp. 1409–1439, Oct. 2007
[12] J. P. H. Wilding et al., “Once-Weekly Semaglutide in Adults with Overweight or Obesity,” N. Engl. J. Med., vol. 384, no. 11, pp. 989–1002, Feb. 2021
[13] Y. P. Silva, A. Bernardi, and R. L. Frozza, “The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication ,” Frontiers in Endocrinology , vol. 11. 2020
[14] C. M. Cuesta, C. Guerri, J. Ureña, and M. Pascual, “Role of Microbiota-Derived Extracellular Vesicles in Gut-Brain Communication,” International Journal of Molecular Sciences , vol. 22, no. 8. 2021
[15] Y. Tsuji et al., “Bile Acid Sequestrant, Sevelamer Ameliorates Hepatic Fibrosis with Reduced Overload of Endogenous Lipopolysaccharide in Experimental Nonalcoholic Steatohepatitis,” Microorganisms, vol. 8, no. 6, p. 925, Jun. 2020
[16] D. J. Sansome, C. Xie, S. Veedfald, M. Horowitz, C. K. Rayner, and T. Wu, “Mechanism of glucose-lowering by metformin in type 2 diabetes: Role of bile acids,” Diabetes, Obes. Metab., vol. 22, no. 2, pp. 141–148, Feb. 2020
[17] J. J. Eloranta and G. A. Kullak-Ublick, “The Role of FXR in Disorders of Bile Acid Homeostasis,” Physiology, vol. 23, no. 5, pp. 286–295, Oct. 2008
[18] K. Ma, P. K. Saha, L. Chan, and D. D. Moore, “Farnesoid X receptor is essential for normal glucose homeostasis,” J. Clin. Invest., vol. 116, no. 4, pp. 1102–1109, Apr. 2006
[19] X. Zhang et al., “Human Gut Microbiota Changes Reveal the Progression of Glucose Intolerance,” PLoS One, vol. 8, no. 8, 2013.
[20] P. D. Cani and W. M. de Vos, “Next-Generation Beneficial Microbes: The Case of Akkermansia muciniphila,” Front. Microbiol., vol. 8, p. 1765, Sep. 2017
[21] D. Zhang et al., “Metformin Alleviates Hepatic Steatosis and Insulin Resistance in a Mouse Model of High-Fat Diet-Induced Nonalcoholic Fatty Liver Disease by Promoting Transcription Factor EB-Dependent Autophagy,” Front. Pharmacol., vol. 12, p. 689111, Jul. 2021
[22] H. R. Bridges, A. J. Y. Jones, M. N. Pollak, and J. Hirst, “Effects of metformin and other biguanides on oxidative phosphorylation in mitochondria,” Biochem. J., vol. 462, no. 3, pp. 475–487, Aug. 2014
[23] M. R. Owen, E. Doran, and A. P. Halestrap, “Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain,” Biochem. J., vol. 348, no. 3, pp. 607–614, 2000
[24] A. K. Madiraju et al., “Metformin suppresses gluconeogenesis by inhibiting mitochondrial glycerophosphate dehydrogenase,” Nature, vol. 510, no. 7506, pp. 542–546, Jun. 2014
[25] S. Herzig and R. J. Shaw, “AMPK: guardian of metabolism and mitochondrial homeostasis,” Nat. Rev. Mol. Cell Biol., vol. 19, no. 2, pp. 121–135, Feb. 2018
[26] C. S. Zhang et al., “Metformin Activates AMPK through the Lysosomal Pathway,” Cell Metab., vol. 24, no. 4, pp. 521–522, 2016
[27] P. A. Lochhead, I. P. Salt, K. S. Walker, D. G. Hardie, and C. Sutherland, “5-aminoimidazole-4-carboxamide riboside mimics the effects of insulin on the expression of the 2 key gluconeogenic genes PEPCK and glucose-6-phosphatase.,” Diabetes, vol. 49, no. 6, pp. 896–903, Jun. 2000
[28] R. A. Miller, Q. Chu, J. Xie, M. Foretz, B. Viollet, and M. J. Birnbaum, “Biguanides suppress hepatic glucagon signalling by decreasing production of cyclic AMP,” Nature, vol. 494, no. 7436, pp. 256–260, 2013
[29] M. Johanns et al., “AMPK antagonizes hepatic glucagon-stimulated cyclic AMP signalling via phosphorylation-induced activation of cyclic nucleotide phosphodiesterase 4B,” Nat. Commun., vol. 7, no. 1, p. 10856, 2016
[30] M. D. Fullerton et al., “Single phosphorylation sites in Acc1 and Acc2 regulate lipid homeostasis and the insulin-sensitizing effects of metformin,” Nat. Med., vol. 19, no. 12, pp. 1649–1654, 2013
[31] K. Zhou et al., “Reduced-Function SLC22A1 Polymorphisms Encoding Organic Cation Transporter 1 and Glycemic Response to Metformin: A GoDARTS Study,” Diabetes, vol. 58, no. 6, pp. 1434–1439, Mar. 2009
[32] C. J. Bailey, C. Wilcock, and J. H. B. Scarpello, “Metformin and the intestine,” Diabetologia, vol. 51, no. 8, p. 1552, 2008
[33] R. S. Hundal et al., “Mechanism by which metformin reduces glucose production in type 2 diabetes,” Diabetes, vol. 49, no. 12, pp. 2063–2069, Dec. 2000
[34] D. B. Shackelford and R. J. Shaw, “The LKB1-AMPK pathway: metabolism and growth control in tumour suppression,” Nat. Rev. Cancer, vol. 9, no. 8, pp. 563–575, Aug. 2009
[35] A. L. G. Notaro and F. T. L. Neto, “The use of metformin in women with polycystic ovary syndrome: an updated review,” J. Assist. Reprod. Genet., vol. 39, no. 3, pp. 573–579, 202
[36] P. Saraei, I. Asadi, M. A. Kakar, and N. Moradi-Kor, “The beneficial effects of metformin on cancer prevention and therapy: a comprehensive review of recent advances,” Cancer Manag. Res., vol. 11, pp. 3295–3313, Apr. 2019
[37] E. Friberg, N. Orsini, C. S. Mantzoros, and A. Wolk, “Diabetes mellitus and risk of endometrial cancer: a meta-analysis,” Diabetologia, vol. 50, no. 7, pp. 1365–1374, 2007
[38] S. M. Samuel, E. Varghese, S. Varghese, and D. Büsselberg, “Challenges and perspectives in the treatment of diabetes associated breast cancer,” Cancer Treat. Rev., vol. 70, pp. 98–111, 2018
[39] Z. Zhu, X. Wang, Z. Shen, Y. Lu, S. Zhong, and C. Xu, “Risk of bladder cancer in patients with diabetes mellitus: an updated meta-analysis of 36 observational studies,” BMC Cancer, vol. 13, no. 1, p. 310, 2013
[40] E. Giovannucci et al., “Diabetes and cancer: a consensus report,” Diabetes Care, vol. 33, no. 7, pp. 1674–1685, Jul. 2010
[41] S. Sośnicki, M. Kapral, and L. Węglarz, “Molecular targets of metformin antitumor action,” Pharmacol. Reports, vol. 68, no. 5, pp. 918–925, 2016
[42] A. Vazquez-Martin, C. Oliveras-Ferraros, S. Cufí, B. Martin-Castillo, and J. A. Menendez, “Metformin activates an Ataxia Telangiectasia Mutated (ATM)/Chk2-regulated DNA damage-like response,” Cell Cycle, vol. 10, no. 9, pp. 1499–1501, May 2011
[43] S. Agarwal, C. M. Bell, S. B. Rothbart, and R. G. Moran, “AMP-activated Protein Kinase (AMPK) Control of mTORC1 Is p53- and TSC2-independent in Pemetrexed-treated Carcinoma Cells,” J. Biol. Chem., vol. 290, no. 46, pp. 27473–27486, Nov. 2015
[44] I. Ben Sahra et al., “Metformin, Independent of AMPK, Induces mTOR Inhibition and Cell-Cycle Arrest through REDD1,” Cancer Res., vol. 71, no. 13, pp. 4366–4372, Jun. 2011
[45] D. M. Sabatini, “Twenty-five years of mTOR: Uncovering the link from nutrients to growth,” Proc. Natl. Acad. Sci. U. S. A., vol. 114, no. 45, pp. 11818–11825, 2017
[46] V. H. Villar et al., “mTORC1 inhibition in cancer cells protects from glutaminolysis-mediated apoptosis during nutrient limitation,” Nat. Commun., vol. 8, p. 14124, Jan. 2017
[47] Y. Yi, W. Zhang, J. Yi, and Z.-X. Xiao, “Role of p53 Family Proteins in Metformin Anti-Cancer Activities,” J. Cancer, vol. 10, no. 11, pp. 2434–2442, May 2019
[48] Y. Han, H. Xie, Y. Liu, P. Gao, X. Yang, and Z. Shen, “Effect of metformin on all-cause and cardiovascular mortality in patients with coronary artery diseases: a systematic review and an updated meta-analysis,” Cardiovasc. Diabetol., vol. 18, no. 1, p. 96, 2019
[49] X. Rossello and D. Yellon, “the risk pathway and beyond,” Basic Res Cardiol, vol. 113, no. 2, 2018.
[50] C. Driver, K. D. S. Bamitale, A. Kazi, M. Olla, N. A. Nyane, and P. M. O. Owira, “Cardioprotective Effects of Metformin,” J. Cardiovasc. Pharmacol., vol. 72, no. 2, 2018
[51] L. K. Seidlmayer, V. V Juettner, S. Kettlewell, E. V Pavlov, L. A. Blatter, and E. N. Dedkova, “Distinct mPTP activation mechanisms in ischaemia–reperfusion: contributions of Ca2+, ROS, pH, and inorganic polyphosphate,” Cardiovasc. Res., vol. 106, no. 2, pp. 237–248, May 2015
[52] Y. Zhang et al., “Metformin and the risk of neurodegenerative diseases in patients with diabetes: A meta-analysis of population-based cohort studies,” Diabet. Med., vol. 39, no. 6, p. e14821, Jun. 2022
[53] G. M. Fahy et al., “Reversal of epigenetic aging and immunosenescent trends in humans,” Aging Cell, vol. 18, no. 6, p. e13028, Dec. 2019
[54] I. Mohammed, M. D. Hollenberg, H. Ding, and C. R. Triggle, “A Critical Review of the Evidence That Metformin Is a Putative Anti-Aging Drug That Enhances Healthspan and Extends Lifespan,” Front. Endocrinol. (Lausanne)., vol. 12, no. August, pp. 1–24, 2021
[55] M. G. Novelle, A. Ali, C. Diéguez, M. Bernier, and R. de Cabo, “Metformin: A hopeful promise in aging research,” Cold Spring Harb. Perspect. Med., vol. 6, no. 3, pp. 1–12, 2016
[56] H. Nasri and M. Rafieian-Kopaei, “Metformin: Current knowledge,” J. Res. Med. Sci., vol. 19, no. 7, pp. 658–664, Jul. 2014, [Online]. Available: https://pubmed.ncbi.nlm.nih.gov/25364368
[57] L. Guo, J. Ma, J. Tang, D. Hu, W. Zhang, and X. Zhao, “Comparative Efficacy and Safety of Metformin, Glyburide, and Insulin in Treating Gestational Diabetes Mellitus: A Meta-Analysis,” J. Diabetes Res., vol. 2019, p. 9804708, 2019
[58] N. Kothari and B. Ganguly, “Potential Drug – Drug Interactions among Medications Prescribed to Hypertensive Patients,” J. Clin. Diagn. Res., vol. 8, no. 11, pp. HC01-HC4, Nov. 2014.
[59] B. A. Kroner, “Common drug pathways and interactions,” Diabetes Spectr., vol. 15, no. 4, p. 249, 2002
[60] S. Steinlauf et al., “Potential drug interactions in travelers with chronic illnesses: A large retrospective cohort study,” Travel Med. Infect. Dis., vol. 12, no. 5, pp. 499–504, 2014
[61] N. M. Pakkir Maideen, A. Jumale, and R. Balasubramaniam, “Drug Interactions of Metformin Involving Drug Transporter Proteins,” Adv. Pharm. Bull., vol. 7, no. 4, pp. 501–505, Dec. 2017
[62] B. Bostrom, P. Uppal, J. Chu, Y. Messinger, L. Gandrud, and R. McEvoy, “Safety and Efficacy of Metformin for Therapy-induced Hyperglycemia in Children With Acute Lymphoblastic Leukemia,” J. Pediatr. Hematol. Oncol., vol. 35, no. 7, 2013
[63] R. A. Vigersky, A. Filmore-Nassar, and A. R. Glass, “Thyrotropin Suppression by Metformin,” J. Clin. Endocrinol. Metab., vol. 91, no. 1, pp. 225–227, Jan. 2006
[64] J. A. Kraut and N. E. Madias, “Metabolic Acidosis of CKD: An Update,” Am. J. Kidney Dis., vol. 67, no. 2, pp. 307–317, 2016



