Key Source of Thymic Aging Discovered
- This research sheds some light on why thymic involution occurs.

Publishing in Nature Communications’ Cell Death Discovery, a team of Chinese researchers has discovered a reason for thymic involution, the age-related decline of the thymus.
The thymus and its involution
The thymus is a boot camp for T cells. Originating from bone marrow, T (for thymus) cells are chemically trained in the thymus, where they learn to attack antigens and not to attack the body’s tissues. However, over time, thymic tissue is gradually replaced by fat tissue, and its function decreases accordingly. This process is known as involution, and combating thymic involution has long been a goal of aging research.
In their introduction, the researchers of this study explain that thymic function is governed by thymic epithelial cells (TECs). When TECs are dysregulated and the function of the thymus falters, this can lead to both infection susceptibility [1] and autoimmune disorders [2].
Dendritic cells, Jagged, and Notch
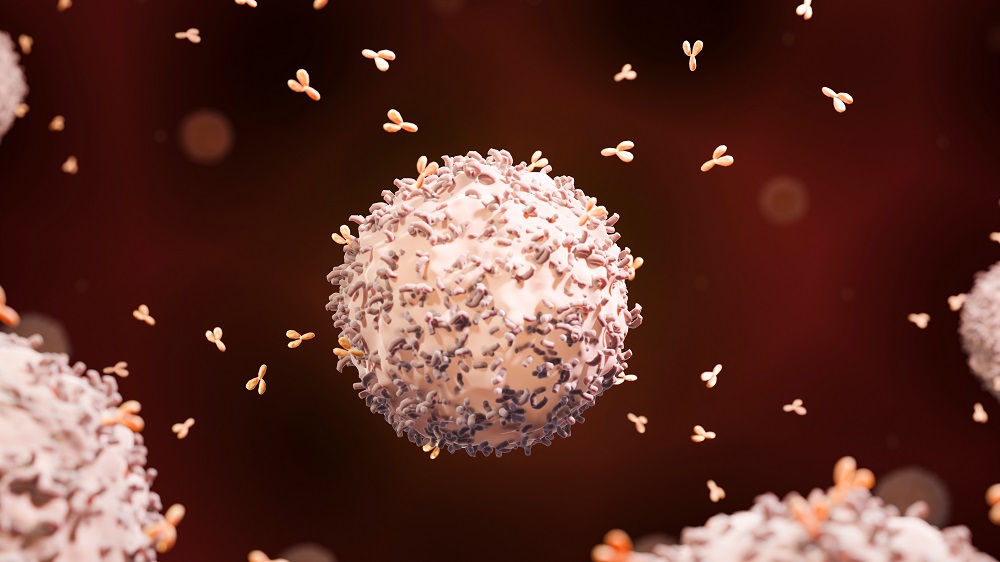
Dendritic cells are part of the adaptive immune system. As the researchers explain, dendritic cells are tissue-resident cells that migrate into lymph nodes and provide multiple necessary functions, including transforming naive T cells into effector T cells. However, these cells can and do enter the thymus itself, where their activity can be negative.
The researchers showed that through direct cell-to-cell communication, dendritic cells express a ligand called Jagged, which binds to the Notch protein. This protein is critical to the proper functioning of the thymus, and this study provides evidence that Notch3 is expressed by TECs. Through this intercellular communication, the dendritic cells activated the Notch signaling pathway in a way that induced cellular death (apoptosis) of the TECs and inhibited their proliferation.
The researchers conducted further experiments to verify their results. Injecting dendritic cells led to thymic atrophy, as did injection of the Jagged ligand.
Abstract
Multiple proinflammatory conditions, including chemotherapy, radiotherapy, transplant rejection, and microbial infections, have been identified to induce involution of the thymus. However, the underlying cellular and molecular mechanisms of these inflammatory conditions inducing apoptosis of thymic epithelial cells (TECs), the main components of the thymus, remain largely unknown. In the circulation, mature dendritic cells (mDCs), the predominant initiator of innate and adaptive immune response, can migrate into the thymus. Herein, we demonstrated that mDCs were able to directly inhibit TECs proliferation and induce their apoptosis by activating the Jagged1/Notch3 signaling pathway. Intrathymic injection of either mDCs or recombinant mouse Jagged1-human Fc fusion protein (rmJagged1-hFc) into mice resulted in acute atrophy of the thymus. Furthermore, DAPT, a ?-secretase inhibitor, reversed the effects induced by mDC or rmJagged1-hFc. These findings suggest that acute or aging-related thymus degeneration can be induced either by mass migration of circulating mDCs in a short period of time or by a few but constantly homing mDCs.
Conclusion
Although this is a basic research study that has not examined any potential treatments, it paves the way for the possible development of signaling-based interventions that use the relationship between Jagged and Notch to slow and/or reverse thymic involution. However, while the researchers have clearly shown the role of Notch signaling as a cause of thymic aging in human beings, they also note that it is not the only cause. These signals are part of an intricate network, so directly inhibiting Notch signaling may or may not be a fruitful endeavor.
Similarly, this research should be looked at in light of treatments that have been reported to restore thymic function, such as the HGH and DHEA treatment championed by Intervene Immune.
Literature
[1] Kellogg, C., & Equils, O. (2021). The role of the thymus in COVID-19 disease severity: implications for antibody treatment and immunization. Human Vaccines & Immunotherapeutics, 17(3), 638-643.
[2] Abramson, J., & Anderson, G. (2017). Thymic epithelial cells. Annual review of immunology, 35, 85-118.