In a preprint paper, scientists have shown that treating drosophila flies and mice with rapamycin for a relatively brief period in early life mostly recapitulates the effects of a lifelong treatment, including lifespan extension in flies [1].
Rapamycin is considered one of the most promising geroprotective drugs. It belongs to a handful of molecules that have shown reproducible lifespan extension in mice, and it is currently undergoing trials in humans and dogs.
When rapamycin was first tested by the Intervention Testing Program (ITP), which is the current gold standard for testing life-prolonging interventions in mice, the researchers were amazed to find that mice who started receiving rapamycin at the respectable age of 20 months benefited from virtually the same increase in lifespan as mice who had been fed rapamycin their entire life [2]. This finding made waves, since it demonstrated that it is possible to intervene late in life and still achieve a considerable lifespan extension.
How much rapamycin is just enough?
Unfortunately, rapamycin also has toxicity issues, so researchers have been searching for the minimum dose required to prolong life. That includes this study, in which the researchers started with drosophila flies and then moved on to mice. Although they are not as close to humans as mice are, drosophila flies, with a maximum lifespan of just over three months, are considered a cheap and effective model for proof-of-concept longevity studies.
The researchers divided the flies into several groups. In one group, the flies were fed rapamycin for their entire lives, which led to an expected increase in lifespan. What was less expected though, is that another group, which contained flies that had been fed rapamycin only during the first 15 days of their lives, enjoyed the same increase in lifespan. The researchers immediately dubbed this phenomenon “rapamycin memory”.
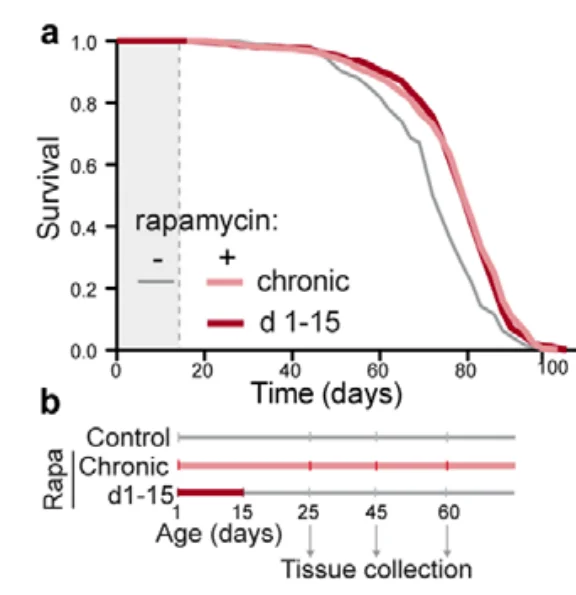
Rapamycin affects nutrient sensing by interacting with the protein complex TOR (target of rapamycin). Basically, rapamycin nudges the organism towards maintenance and away from growth, which is why rapamycin treatment is known do decrease the number of actively dividing cells.
Drosophila guts are characterized by high rates of cellular division and turnover (the replacement of damaged cells by intestinal stem cells). This drives intestinal dysplasia (the presence of abnormal cells, which is considered a pre-cancerous condition) and shortens lifespan. In both groups, the rapamycin treatment resulted in reduced turnover, as cells remained healthy for longer, and there was less need to replace them.
This translated to fewer intestinal pathologies as well. While in the controls, aging resulted in widespread dysplasia, flies in both treatment groups had significantly fewer dysplastic lesions throughout their lifespans. Rapamycin also helped preserve gut barrier function; it protected the flies from leaky gut, a major cause of aging in both flies and humans [3].
It’s the autophagy
How exactly does “rapamycin memory” work? While the activity of target of rapamycin complex 1 (TORC1) was reduced by the treatment, it bounced back just 48 hours after withdrawal. This means that the repression of TORC1 by rapamycin must have activated other mechanisms that remained in place to maintain “rapamycin memory”.
One such downstream consequence of TORC1 repression is increased autophagy, which is required for rapamycin-induced lifespan extension. If autophagy remained upregulated even after the withdrawal of rapamycin, this could explain the “rapamycin memory” effect, and this is exactly what the researchers found. Even after the brief early-life rapamycin treatment, autophagy levels remained high throughout the flies’ lifespan, although not in the head, which hints at a tissue-specific response.
When the researchers downregulated autophagy by other means, it mostly blocked both rapamycin-induced lifespan extension and improvement in gut health, completely abolishing the “rapamycin memory” effect. On the contrary, when researchers upregulated autophagy for the same short period of time early in life, this caused roughly the same lifespan extension as rapamycin. Notably, the combination of rapamycin and autophagy inhibition fared no better than any single treatment.
Translatable to mammals
To see if “rapamycin memory” translates to mammals, the researchers assessed the effects of early-life short-term rapamycin treatment in mice. The levels of lipopolysaccharide-binding protein (LBP), a marker of bacterial translocation from the intestine into the bloodstream, were kept equally low by both the lifelong and the short-term rapamycin treatments, showing that the latter leads to a lasting improvement in gut health. This was confirmed by analyzing intestinal structure and cellular health in the mice’s guts. The researchers conclude:
Our findings suggest that the full geroprotective effects of rapamycin can be achieved without the adverse effects sometimes seen with chronic, long-term dosing.
Interestingly, one recent study did find that early-life treatment with rapamycin extends lifespan in mice.
Conclusion
Years ago, the scientific community was stunned by the discovery that rapamycin treatment gives the same boost to murine lifespan when started later in life. In this new study, the researchers show that rapamycin works just as well in a brief early-life intervention and suggest a mechanism behind this effect. Given rapamycin’s toxicity record, this finding could help devise better treatment regimens. On the other hand, this study suggests that we might not need rapamycin or similar compounds at all, as upregulating autophagy by other means could potentially recapitulate its effect.
Literature
[1] Juricic, P., Lu, Y. X., Leech, T., Drews, L. F., Paulitz, J., Lu, J., … & Partridge, L. (2022). Full geroprotection from brief rapamycin treatment by persistently increased intestinal autophagy. bioRxiv.
[2] Harrison, D. E., Strong, R., Sharp, Z. D., Nelson, J. F., Astle, C. M., Flurkey, K., … & Miller, R. A. (2009). Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. nature, 460(7253), 392-395.
[3] Mishra, S. P., Wang, B., Wang, S., Nagpal, R., Miller, B., Jain, S., … & Yadav, H. (2021). Microbiota induces aging-related leaky gut and inflammation by dampening mucin barriers and butyrate-FFAR2/3 signaling. bioRxiv.





