Lipofuscin is the longest-known but in many ways least-understood intracellular aggregate in human degenerative aging. This hodgepodge of damaged organelles, proteins, lipids, a small amount of carbohydrates, and traces of metals accumulates in critical long-lived cell types like brain neurons and heart and skeletal muscle cells.
But despite having known about it for more than a hundred years, scientists still don’t know very much about how lipofuscin is formed, or exactly from what, or what its molecular composition is. Lipofuscin fluoresces across a wide range of the spectrum, which suggests its composition is heterogenous — and those constituents are heavily crosslinked within and amongst each other, which makes it impossible to identify individual constituents, even when using fairly high-tech methods like mass spectroscopy. It’s generally agreed that incompletely-degraded mitochondria are one important constituent of lipofuscin — but this impression may mostly arise from the fact that scientists’ tools can readily pick up their molecular signature, while everything else in lipofuscin degenerates into an inchoate goo.
Whatever its origin, lipofuscin seems to grow once formed, probably because its surface is reactive and thus snags onto everything with which it comes in contact, like the Blob from the campy 1950s horror flick. One extreme example: German scientists found that lipofuscin occupies as much as 70% of the cellular volume of centenarians’ motor neurons!
Why the difference? There’s no way to be sure what was happening in the earlier positive studies, but the investigators pointed to several limitations to the lab techniques used by the 1970s scientists as possible sources of experimental artifact — limitations to which their own study using modern techniques was not vulnerable.
Enough of these feeble or illusory solutions. What would it take to really deal with the stubborn, toxic waste products that accumulate in our cells with age?
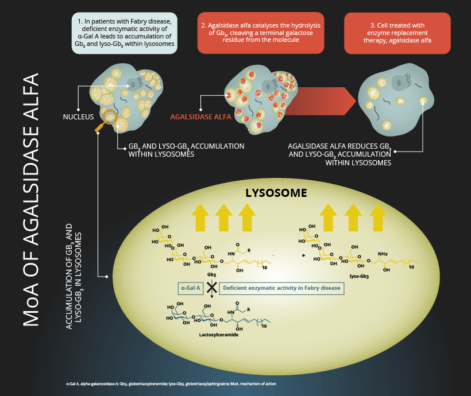
Instead of replacing enzymes that most people are born with in people who genetically lack them, many potential LysoSENS rejuvenation biotechnologies are forms of ERT that deliver entirely new enzymes to target cells. Such enzymes would be engineered to break down wastes that no one’s lysosomes are currently able to process, and that therefore accumulate in our cells with age and cause dysfunction and disease.
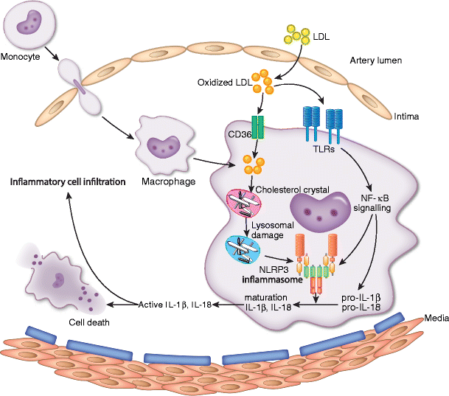
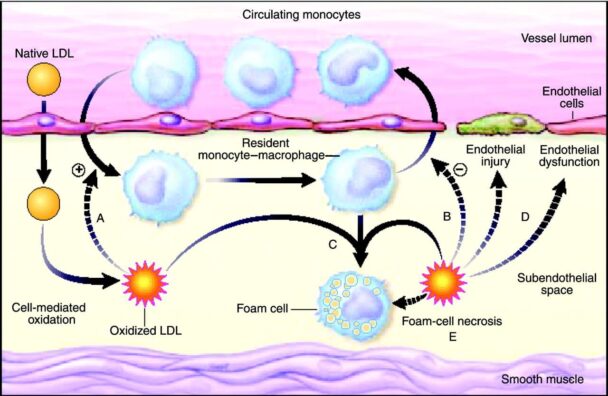
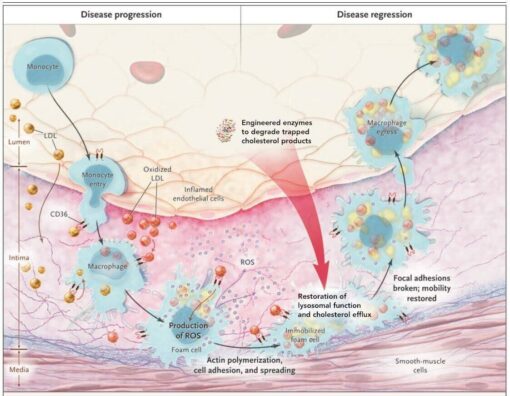
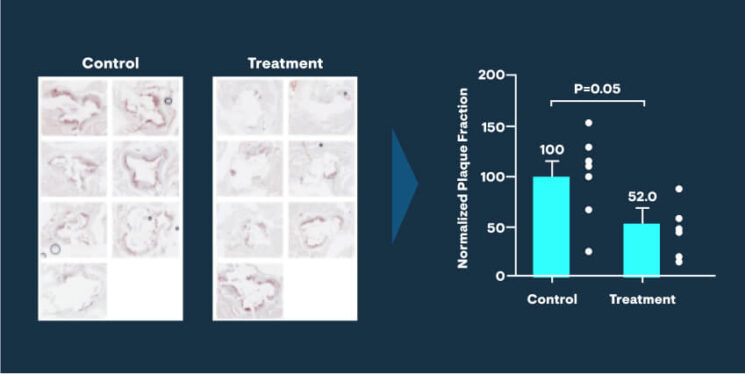
For example, enzymes capable of breaking down the toxic cholesterol byproducts responsible for foam cell formation would give superpowers to macrophages. Such enzymes would rehabilitate existing foam cells, allowing them to mobilize safely out of atherosclerotic lesions, and healthy enzyme-enhanced macrophages would be able to remove toxic cholesterol products from the artery wall as well as dead and dying foam cells without harm to themselves. Nascent atherosclerotic plaques would not progress, and existing ones would regress.
Another engineered enzyme would remove A2E and related compounds from the RPE cells in the eye, which would arrest and reverse RPE cell loss; macular degeneration would be prevented, and its early stages reversed.
A different approach is needed to clear out aggregates that accumulate inside aging cells but outside of the lysosome. Examples include aberrant forms of the proteins tau and alpha-synuclein, which accumulate inside neurons and are drivers of key aspects of Alzheimer’s and Parkinson’s as well as other neurological disorders of aging, including the so-called “normal” cognitive and motor decline of aging that doesn’t reach the “disease” threshold in a given person.
It’s not always clear why these aggregates aren’t sent to the lysosome and degraded. Some kinds of aggregate may only build up in the main body of the cell once the lysosome begins to fail with age due to the accumulation of those aggregates that are sent to the lysosome but not successfully degraded. Such lysosomes are known to become dysfunctional and to stop accepting delivery of new materials for degradation, which in this case might include the precursors of the aggregates that tend to accumulate with age outside of the neurons, reminiscent of the kind of logistical snarls that have bedeviled supply chains as the industrialized world has emerged from the pandemic. In cases where this is the core problem, it should clear up once we purge aging lysosomes of these other, primary kinds of waste, freeing them up to once again accept aggregates that currently sit outside the lysosome like so many container ships adrift in the waters surrounding the Port of Long Beach.
However, some of the intracellular aggregates that accumulate in the main body of the cell instead of in the lysosome them may not be targeted to the lysosome in the first place, because our cells have never evolved machinery to drag them there. Such cases would require novel strategies for their more direct removal, which is a tricky business because it would have to be done without the protective membrane and otherwise-toxic concentrations of acid and powerful enzymes that make the lysosome so effective at degrading damaged molecules. As we will discuss below, a strategy to potentially handle such cases has only recently emerged.