Scientists from Johns Hopkins Medicine have reported the successful creation of a special type of gel that mimics the lymph nodes in our bodies. This gel recruits and multiplies T cells just like actual lymph nodes do, so it could help in the fight against cancer and immune system disorders.
The lymph nodes are the boot camps of the immune system
There has been a great deal of interest in immunotherapy in the last few years, particuarly in using the T cells, a type of white blood cell, to hunt down cancer and destroy it. Our own immune system is quite literally living medicine, and when it works properly, it can deal with invading pathogens and cancers with ease; this is one reason why the traditional small-molecule approach to cancer has started to fall by the wayside in favor of immune approaches.
Immunotherapy typically increases the amounts of immune cells available or even boosts how well the cells work in order to fight off disease. Cancer often hides in plain sight and uses various tricks to fool the immune system into ignoring it, so immunotherapy often focuses on improving how well our immune cells detect cancer or help it to see past the tricks it uses to hide.
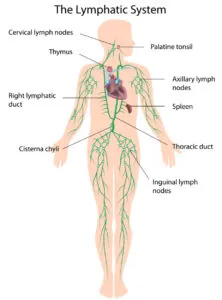 In order to do this, the immune cells must first be trained to detect the key molecular markings on the surfaces of cancer cells. This normally happens in the lymph nodes, which function almost like army boot camps where the new T cell recruits are trained for battle; this follows their initial training in the thymus.
In order to do this, the immune cells must first be trained to detect the key molecular markings on the surfaces of cancer cells. This normally happens in the lymph nodes, which function almost like army boot camps where the new T cell recruits are trained for battle; this follows their initial training in the thymus.
The lymph nodes are small, bead-like glands that are distributed around the body in order to provide a comprehensive network of defenses. In patients with immune system disorders or cancer, this training does not happen or is incomplete, so the resulting T cells cannot do their job properly. With aging, the lymph nodes become increasingly dysfunctional as they become fibrotic and deteriorate.
Currently, the solution to this lack of training is to take the T cells from the patient and activate them through genetic engineering or drugs that complete the training so that the cells can identify cancer cell surface markers properly. This is, by its nature, challenging and costly to undertake and only possible through specialized labs, and it can take several weeks to do. Furthermore, once the T cells are given back to the patient, they do not last long before more need to be processed in this way.
Moving towards an artificial lymph node
In the new study, researchers developed a type of hydrogel, a jelly-like polymer, to serve as a platform for T cell training and which mimics the lymph node environment [1]. The hydrogel contains two chemical signals that encourage T cells to locate and eliminate foreign targets, and it acts similarly to real lymph nodes in training these cells. Compared to the T cells activated in standard Petri dishes, the cells on the hydrogels produced an impressive 50 percent more activation cytokines.
The team also experimented with different grades of hydrogels, ranging from very soft consistency to harder more rigid ones. They found that the T cells favored a softer gel environment over the more tightly packed rigid gels. Over 80 percent of the T cells in the soft gel divided, while those in the rigid gel did not.
To put this into perspective, this translates to taking a handful of cells and having them multiply to around 150,000 cells in just a week. The team also compared this with the Petri dish method of activating and multiplying T cells, which only reached around 20,000 cells in the same period. This makes this gel approach much more efficient, and the potential price point of therapy could be considerably lower with this method.
Finally, the researchers studied mice with a lethal form of melanoma and injected them with T cells from both the hydrogel and the regular Petri dish cultures. The tumors in the mice given gel-derived T cells stabilized and did not grow in size, while an injection of regular Petri dish T cells led to continued tumor growth in most of the mice.
Abstract
T cell therapies require the removal and culture of T cells ex vivo to expand several thousand‐fold. However, these cells often lose the phenotype and cytotoxic functionality for mediating effective therapeutic responses. The extracellular matrix (ECM) has been used to preserve and augment cell phenotype; however, it has not been applied to cellular immunotherapies. Here, a hyaluronic acid (HA)‐based hydrogel is engineered to present the two stimulatory signals required for T‐cell activation—termed an artificial T‐cell stimulating matrix (aTM). It is found that biophysical properties of the aTM—stimulatory ligand density, stiffness, and ECM proteins—potentiate T cell signaling and skew phenotype of both murine and human T cells. Importantly, the combination of the ECM environment and mechanically sensitive TCR signaling from the aTM results in a rapid and robust expansion of rare, antigen‐specific CD8+ T cells. Adoptive transfer of these tumor‐specific cells significantly suppresses tumor growth and improves animal survival compared with T cells stimulated by traditional methods. Beyond immediate immunotherapeutic applications, demonstrating the environment influences the cellular therapeutic product delineates the importance of the ECM and provides a case study of how to engineer ECM‐mimetic materials for therapeutic immune stimulation in the future.
Conclusion
This early, proof-of-concept work is one step closer towards injecting these artificial gel lymph nodes into people who would benefit from an improved level of immune system response, and it opens the door to fighting cancer and other diseases. The study highlights the importance of the substrate on which cells are placed in influencing them and thus the importance of the extracellular matrix (ECM).
The researchers discuss how this study not only improves current immunotherapy cell expansion methods but also lays the groundwork for creating ECM-mimetic materials for therapeutic immune stimulation in the future. This is only an initial step towards creating lymph nodes, but studies like this demonstrate the concept’s plausibility and so bring such things closer to becoming a reality.
Literature
[1] Hickey, J. W., Dong, Y., Chung, J. W., Salathe, S. F., Pruitt, H. C., Li, X., … & Gerecht, S. (2019). Engineering an Artificial T‐Cell Stimulating Matrix for Immunotherapy. Advanced Materials, 1807359.